- The temperature, the equivalence ratio and the residence time in the primary zone of the combustor (PZ).
- The degree of homogeneity of the primary-zone combustion process.
- The quenching characteristic of the liner’s walls.
- The zones downstream of the PZ. Although the effect of these zones may be positive in terms of carbon monoxide (CO), unburnt hydrocarbons (UHC) and soot oxidation, in some cases, quenching effects that take place within these zones may result in “freezing” of the above pollutant concentrations before the combustion is completed. The latter is of significant importance in designing fuel-rich primary zones.
The most important factors that affect CO emission concentrations are:
- CO is produced in all fuel/air ratios.
- At low temperatures, that corresponding to low fuel/air ratios (Φpz < 0.5), CO cannot be burnt to CO2.
- At high temperatures or high fuel/air ratios (Φpz > 0.9) on the other hand, CO2 dissociates to CO.
- Improved fuel atomization. It promotes fuel evaporation and enhances the mixing process, so as to achieve a more homogenous state. This can be done by using airblast instead of pressure atomizers. Another approach is to use air assistance nozzles in order to improve atomization at low fuel flows.
- Holding the primary-zone equivalence ratio to its optimum value (0.7) by redistributing the air flow.
- Increase the residence time or the primary’s zone volume.
- Reduce the film cooling air. In the figure below the negative effect of film cooling air in CO emissions is shown. This occurs because the air from the PZ usually contains high concentrations of CO and UHC, which due to the film-cooling air are “freezing” and appear in the exhaust gas. So, a more effective cooling system that requires less cooling air will have a positive effect of CO reduction.
- Use of compressed air-bleed. In this approach air is bled from the compressor at low power operation, which increases the fuel/air ratio and the temperature of the primary zone, whereas reduces the CO emissions.
- Use of fuel staging. In this technique the fuel supply in some nozzles is cut and diverted to the remainder. At low power conditions this method is able to reduce CO emissions by increasing the local FAR in the burning zone and improving atomization quality.
Unburnt hydrocarbons
The parameters that influence CO emissions are also affecting UHC emissions in the same way. So, UHC emissions reduction requires the same treatment with CO emissions, but more attention should be paid on the reduction of film-cooling air in the primary zone, as well as on fuel atomization improvements. The latter could be achieved with the use of airblast atomizers and air assist nozzles.
Oxides of nitrogen
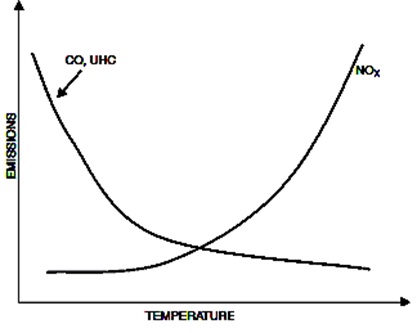
The principal parameter that governs NOx formation is temperature. All the other combustor parameters should be regarded as important only if affecting the flame temperature. Thus, the prime goal of any NOx reduction technique is the decrement of flame temperature, which as it is shown in the figure below, is exponentially related with NOx emissions.
Another important parameter should be the “hot spot” elimination from the reaction zone. “Hot spots” are localized regions of high temperature where NOx formation is high, although the mean temperature in the reaction zone may be sufficiently low. Furthermore, the time that is available for NOx formation must be kept close to a minimum value.
An increase in the airflow of the primary zone will decrease both flame temperature and residence time. Although this will reduce NOx emissions, it will increase CO and UHC emissions as it is shown in the following figure. Unfortunately, almost in all techniques that are used for emissions reduction there is a great deal of compromise between CO and UHC on one hand and NOx and smoke on the other.
In order to develop “low NOx” combustors modifications must be done in the conventional design. For instance, changes in the distribution of air and liner’s geometry, more sophisticated techniques of fuel injection, new wall-cooling methods should be developed. The advantage of this approach is that the combustor maintains its existing size and configuration, while the improvements are within the bounds of established technology. The main drawback is that the final combustor will inevitably be a compromise between emissions and other performance characteristics.
The various approaches of NOx reduction in conventional combustion chambers involve some of the following:
- Lean primary zone. It was a common practice in the past since apart from NOx reduction lean mixtures could minimize combustor’s size and make the ignition easier. However, the excess of air reduces the flame temperature, having as a consequence augmented CO and UHC emissions. Therefore, the NOx reduction that can be achieved is limited.
- Rich primary zone. It is the exact opposite approach. The idea is to use excess of fuel and air in order to produce lower flame temperature and consequently less NOx emissions. The main drawback of this technique is the necessity of fuel-rich products passing through the stoichiometric state very rapidly, so as to reach turbine inlet conditions. Thus, the limitation in this case is that the time between fuel-rich and fuel-lean conversion should be sufficient for CO and UHC oxidation.
- Improved combustion’s homogeneity. If the mixing rate is improved prior to the combustor it will reduce the range of flame temperature. If this is combined with a lean fuel/air ratio NOx will be reduced, but, if the average fuel/air ratio is stoichiometric, NOx will be increased.
- Residence time’s decrement. By reducing the time that gases spend in the high combustor temperatures, NOx emissions will be diminished. However, this has a negative effect on CO and UHC emissions, since there is no sufficient time for their oxidation.
- Use of water injection. NOx formation is highly dependent on temperature. Thus, the injection of an inert, non-combustible substance, such as water, which can dilute the fuel/air mixture, will result in a considerable NOx reduction. Direct water injection was first used for smoke reduction. Its main drawbacks are related with the problems involved in pumping, storing and handling the large amounts of demineralized water required for its operation. This is the reason why is used mainly in large stationary industrial gas turbines.
- Recirculation of exhaust gas. Exhaust gas is another inert substance that is available in abundance, but in order to be effectively used for NOx reduction, it should be cooled first before it re-enters to the primary zone. Although this technique can sufficiently reduce NOx emissions, sometimes there is a large increase in CO emissions. Furthermore, the recirculation demands an increased size, weight and complexity of the combustion equipment.
Soot – smoke
In many practical systems soot formation is governed by the physical processes of fuel atomization and fuel-air mixing, rather than by chemical kinetics. The general idea in smoke reduction is to prevent the occurrence of fuel-rich pockets in the flame. The injection of additional air in the primary zone is beneficial, especially if it is combined with an increase in the liner’s pressure drop. The combination of more oxygen, lower temperature and better mixing can significantly decrease soot formation and consequently smoke. However, this approach is restricted because apart from the desired smoke reduction, there are some adverse effects on ignition and stability limits, as well as an increase in CO and UHC emissions at idle operation.
According to Villasenor and Kennedy [3], soot may be a significant sink of OH and may lead to local extinction of the reaction zone near the flame tip. A simple approach to reduce smoke is to replace the pressure-type atomizers with airblast atomizers, which can considerably reduce smoke due to the better atomization and aeration of the fuel that they achieve.
Another method for smoke depletion is water injection (discussed above). Furthermore, fuel additives have been used in the past in order to reduce soot with varying success. These additives are consisted of several organo-metallic compounds, which have as a base usually barium and manganese. The main disadvantages of fuel additives are their tendency to produce deposits on the turbine blades, their cost and the risk of introducing a toxic hazard.
References
The article was based on my M.Sc. thesis:
Samaras C., 2010, “Emissions estimation from industrial gas turbine combustors”, M.Sc. Thesis, Cranfield University, UK.
The figures were found from:
[1] Lefebvre, A. H., 1998, “Gas turbine combustion”, Taylor and Francis, 2nd Edition, London, UK.
[2] Boyce, P. M, 2002, “Gas turbine engineering handbook”, Gulf Professional Publishing, 2nd edition, Huston, Texas, USA.
[3] Villasenor, R., and Kennedy, I. M., 1992, “Soot formation and oxidation in laminar diffusion flames”, Twenty-Fourth Symposium (International) on Combustion/The Combustion Institute, Sydney, Australia.
Read also
Emissions Formation In Gas Turbine Combustors
Gas Turbine Combustor Concepts For Low Pollutant Emissions